 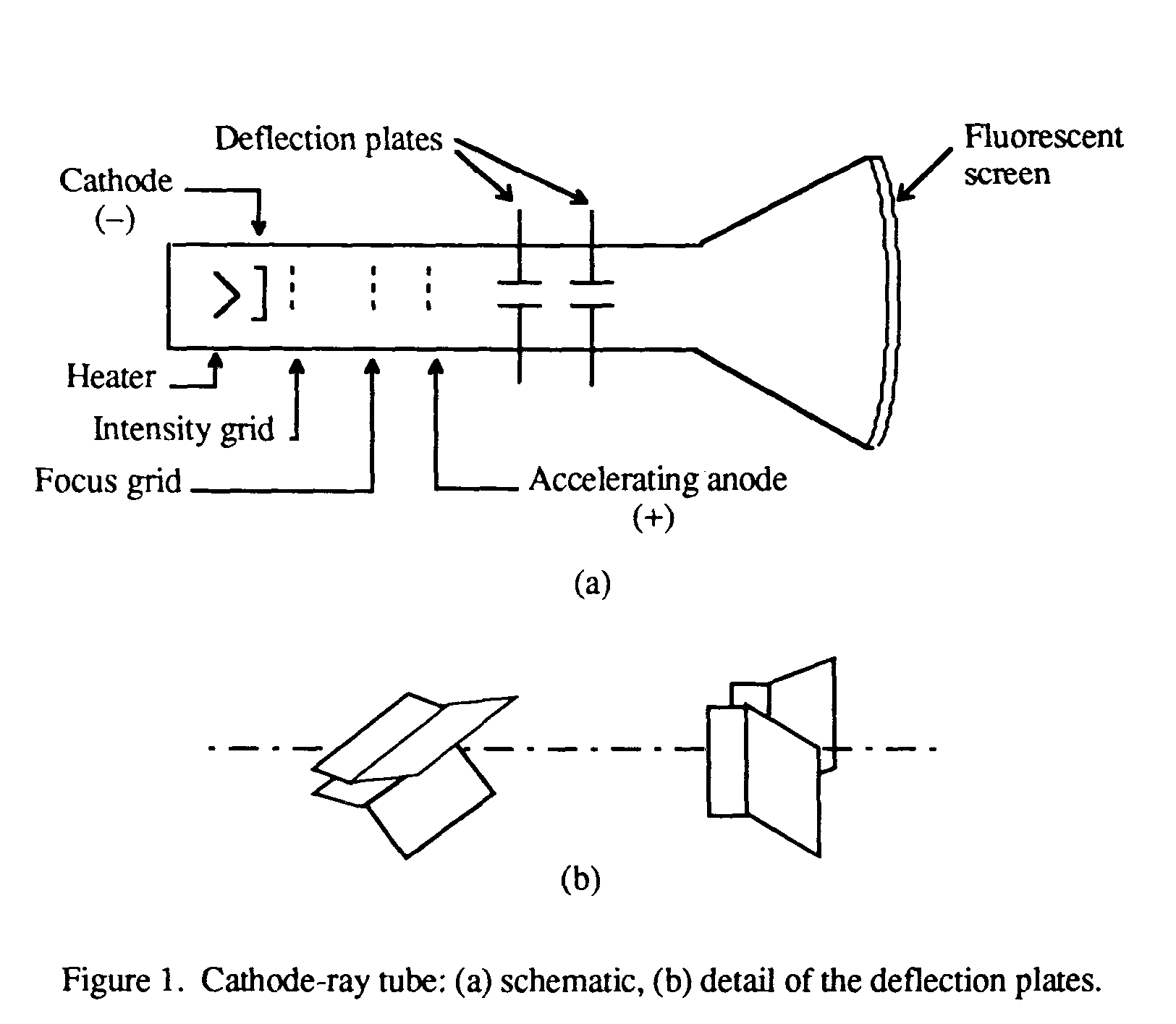
A TV CRT has an electron gun in the back that shoots a beam of electrons at a phosphor screen. These cathode rays are able to be used in televisions because they can be effected by magnetic fields. You can read more about his experiment at this website: 
It was this property of the cathode ray that let JJ Thompson discover and prove the existence of the electron as a subatomic unit of an atom in 1897. This allows them to be manipulated by a magnetic field. The reason that you can bend this ray with a magnet is because electrons are negative. This is why the beam has a blueish purple glow. Because it cannot stay at this high energy state for long, the atom releases this energy as light. When the electrons in the ray hit an oxygen atom, it moves to a higher energy state. Because the vacuum is not perfect, there will still be air molecules in the tube. If you remove most of the air in the chamber though, then the electrons are able to flow from one electrode to another. These electrons want to flow to the anode to equalize the voltage, but cannot because there are too many air molecules blocking their way. The cathode, because it has a negative high voltage with respect to the anode, will have a large amount of electrons on it. Now the reason this ray forms is because of the high voltage. If you draw a vacuum on the chamber and apply a high voltage to both ends, then a blueish purple glowing line forms. The simplest cathode ray tube is a sealed glass chamber that has electrodes at either end and a port to be able to draw a vacuum from. So many of you may be wondering now, how does a cathode ray tube work?Ī cathode ray tube consists of a ray going from the cathode to the anode, and it is made up of electrons. They are still used widely today as oscilloscope screens, but these cathode ray tubes as screens are a little bit more complicated than the simple cathode ray tube that we are going to be building in this instructable. These tubes have been used as television screens for all of the 20th century and for the early part of the 21st century. In fact, the first one was built in 1897 by scientist Ferdinand Braun. The video below compliments this instructable with a visual demonstration of this Cathode Ray Tube in action.ĬRT's have been around for a long time. This piece of test equipment that I will show you how to build can be used as an awesome scientific demonstration for any physics and chemistry class, or it can be used just as a way to confirm a long standing scientific principal. I'll explain how it proves this theory later in this instructable. This line not only looks cool, but it can be used to prove a scientific theory electrons are a subatomic unit with a negative charge. You can manipulate and bend this stream of electrons with a magnet. 
This is a stream of electrons that looks like a brilliant blue-purple line of plasma. When there is a vacuum drawn and a high voltage across the two electrodes, a cathode ray forms. 
A cathode ray tube, or CRT, is a glass tube with electrodes on either end. Luckily, there is a device that will let you. The charge carried by a single proton/electron is the smallest charge carried by any particle.Electrons: they are all around us, but we cannot really see or interact with them. Hydrogen is the lightest element and gives smallest positive particles and is known as proton. Actually these positively charged constituents of the anode rays are nothing but the atoms of the gas left after one or more electrons are knocked out of them. These rays carry positive charged particles and their e/m ratio changes from gas to gas unlike the e/m ratio of the constituent particles of cathode rays. These rays carry a positive charge and hence also known as positive rays. These rays after emitted by anode pass through the pores made in the cathode surface. When careful experiments were carried out using perforated cathode in the discharge tube, a new type of rays known as anode rays were revealed. JEE Main 2022 Question Paper Live Discussion.Difference Between Selling And Marketing.TS Grewal Solutions Class 11 Accountancy.TS Grewal Solutions Class 12 Accountancy.CBSE Previous Year Question Papers Class 12.CBSE Previous Year Question Papers Class 10.NCERT Solutions For Class 6 Social Science.NCERT Solutions for Class 7 Social Science.NCERT Solutions for Class 8 Social Science. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
